I have recently found myself in a number of discussions regarding what it is actually possible to know about measurement accuracy, especially as it relates to sterile compounding. These discussions have ranged from considerations of the lowest volume a human can measure, to the level of precision necessary in density data for gravimetric systems. Based on those conversations, I have come to the conclusion that we tend to practice as if we were capable of more precision and more accuracy than is, in fact, humanly possible in the general case.
- I have observed computation of a weight-based dose of chemotherapy to be 50.2 mL of a drug and seen the pharmacist draw up 50 mL in a 50 mL syringe, and 0.2 mL of the drug in a 1 mL syringe
- I have observed measurements in 1 mL syringes that go down to 0.02 mL.
- I have observed people struggling to pull the plunger on a 2 mL syringe to 1.158 mL
None of these activities achieved what the pharmacist thought they were achieving.
So this is a rant about what reality looks like when we try to measure medications.
Significant figures
Those of us who remember our basic high school chemistry may recall that we were introduced to the concept of something called ‘significant figures’. There were two fundamental lessons from that concept:
- The accuracy of any measurement system is limited by the least accurate technique used in that measurement (you can’t measure millimeters with a yardstick)
- You can’t make a measurement more precise by performing arithmetic on it (you can’t weigh 1000 items on a scale accurate to the nearest gram and produce an accurate weight to the microgram level).
What this, in turn, should teach us is that the accuracy with which we prepare a dose depends entirely on the tools we use to physically measure the compounds that go into the dose. For IV admixtures, these consist of sterile disposable syringes, vials of drugs, and IV bags.
My observation is that we tend to treat these objects as if what appears on their labeling (including the measurement scale on the syringe) was completely and quintessentially accurate. The truth is that it may not be.
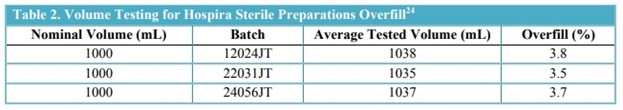
IV Bag Overfill
IV bags have overfill. The amount of overfill is a range, not a number. While one can attempt over time to create an average overfill, it will not likely be correct to the milliliter in any particular instance.
Baxter Overfill Chart

Hospira Overfill Chart
Syringe Accuracy
There is an ISO standard (ISO 7886-1 (2017)) that describes acceptable accuracy of a sterile disposable syringe. To get the whole thing you have to purchase it. The short story here is:
- The most accurate syringe in common use has an accuracy of ±4% when its nominal volume is 5 mL or greater when delivering at 50% or more of its nominal volume.
- This means that (for example), if we fill a 10 mL syringe to its 10 mL mark and we all agree the syringe is full (contains no significant air), the best we can know is that the syringe contains somewhere between 9.6 mL and 10.4 mL.
- This means that (for example), if we fill a 50 mL syringe to its 50 mL mark and we all agree the syringe is full (contains no significant air), the best we can know is that the syringe contains somewhere between 48 and 52 mL.
- Syringes below 5 mL are accurate if they can deliver within ±5% when measuring 50% or more of their nominal volume. For a 1 mL syringe, this means that we know, at best that measuring a 1 mL volume in that syringe will deliver somewhere between 0.95 and 1.05 mL.
- Below 50% of its nominal volume, the accuracy of the syringe declines with the volume measured until, at 10% of its nominal volume, it delivers ±16% accuracy. So when measuring a volume of 0.1 mL in a 1 mL syringe, the best you can actually know is that you are delivering somewhere between 0.084 and 0.116 mL. At 20% of its nominal volume, such a syringe delivers ±9.5% of its apparent measured volume, so, if we were to set ±10% as our acceptable standard of accuracy, then the lowest volume we can accurately measure with any syringe available to us is 0.2 mL
Of course, these numbers are ranges, and any given syringe may be anywhere within this range. These numbers presume measurement to a gradation on the syringe scale. Interpolation between those gradations creates additional error. Further, the inherent error in the performance of the syringe itself makes such interpolation nonsensical. For example, interpolating between the 9.8 mark and the 10.0 mark on a 10 mL syringe, even correctly positioned, only places you somewhere between 9.5 and 10.3 mL.
Densities and Gravimetrics
This has implications for the way we use density to compute volume from changes in mass. As the volume increases, the effect of density precision may become more pronounced, but, at the end of the day, a difference in density only becomes practically useful when that difference describes a difference in volume that we can practically measure with acceptable accuracy. Proposing that a user measure 9.9 mL on a 10 mL syringe is not practical.
Even at two places behind the decimal (e.g. 1.04 vs 1.05), the difference in computed volume is 1%, which is well within the ±4% precision of the syringe. The following table shows the difference in computed volume delivered as density changes from a baseline 1.04, to 1.045 and then to 1.05 for a variety of measurement volumes. The mass of the measurement at baseline 1.04 gm/mL to the volume that same mass would represent at 1.045 and 1.05 mL as shown in the following table.
|
Volume
|
1.045
|
1.05
|
Difference
At 1.045
|
Difference at 1.05
|
Syringe Graduation
|
Minimum Accurate Measurement
(20% nom vol)
|
|
1 mL
|
0.995 mL
|
0.99 mL
|
0.005 mL
|
0.01 mL
|
0.02 mL
|
0.2 mL
|
|
2 mL
|
1.99 mL
|
1.98 mL
|
0.01 mL
|
0.02 mL
|
0.2 mL
|
0.4 mL
|
|
3 mL
|
2.99 mL
|
2.97 mL
|
0.014 mL
|
0.03 mL
|
0.2 mL
|
0.6 mL
|
|
5 mL
|
4.98 mL
|
4.95 mL
|
0.024 mL
|
0.05 mL
|
0.2 mL
|
1 mL
|
|
10 mL
|
9.948 mL
|
9.905 mL
|
0.048 mL
|
0.095 mL
|
0.2 mL
|
2 mL
|
|
20 mL
|
19.904 mL
|
19.81 mL
|
0.096 mL
|
0.19 mL
|
1 mL
|
4 mL
|
|
30 mL
|
29.855 mL
|
29.714 mL
|
0.144 mL
|
0.286 mL
|
1 mL
|
6 mL
|
|
50 mL
|
49.761 mL
|
49.524 mL
|
0.239 mL
|
0.476 mL
|
2 mL
|
10 mL
|
The short story here is that even the difference between two proximate values at two points behind the decimal (e.g. 1.04 and 1.05), results in a change in volume that cannot be practically measured, much less be accurately (within ±10%) by a syringe that can measure the entire volume.
Products used in TPN compounding have contained density information (as specific gravity) for many years to two places behind the decimal without apparently needing to change them on a lot by lot basis. Examples include varying concentrations of Dextrose, and concentrated sodium chloride.
Commercial medication fill accuracy
The amount of medication in a source container of an injection is defined by the USP monograph for that injection. For example, according to the monograph for Cefazolin Sodium for Injection, a container is considered accurately filled if it contains between -10% and +15% of its labeled potency. Based on looking at over 250 monographs of liquid injections, most are within ±10% (though there are some significant outliers. Again, this doesn’t mean that all instances of any product are at these limits; it does mean that any instance of a product could be at these limits and be considered to be accurately filled.
The tolerance stack
The engineering world has a useful concept called a ‘tolerance stack’ that assesses the impact of the accumulation of all the known tolerances within a system to determine the variance within that system. Let’s apply that analysis to the delivery of 1 gram of Cefazolin Sodium from a commercial drug vial into a 50 mL IV piggyback bag:
- A vial labeled to contain 1 gram of Cefazolin Sodium (as described in its USP monograph) is -10% to +15%. While it is likely that not all vials fall at the ends of this range, the best we can know is that the vial itself contains somewhere between 900 mg and 1,150 mg. It seems likely that vendors generally err on the heavy side of that equation, but the likelihood that a vial contains exactly 1 gram is small.
- At least some vials of Cefazolin 1 gram indicate that reconstitution with 9.6 mL of a suitable diluent produces a solution 10 mL of which contains 1 gram (100 mg/mL). Presuming a completely accurate delivery of that diluent (or, as our European colleagues call it, the solvent), that means that we know that we have somewhere between 90 mg/mL and 115 mg/mL concentration.
- The reality from ISO7886-1, however, is that delivering 9.6 mL from a 10 mL syringe has a ±4% precision, which means that we only know for certain that we delivered somewhere between 9.22 and 9.98 mL of diluent. Presuming the expansion from 9.6 to 10 mL is roughly linear within this narrow range, that means that our resulting volume is somewhere between 9.6 and 10.4 mL, so our concentration is somewhere between 86.5 mg/mL (900 mg/10.4 mL) and 119.8 mg/mL (1,150 mg/9.6 mL).
- Pulling a dose back out of that vial (presuming we measure it, rather than just pulling the vial dry), we again use a 10 mL syringe with a ±4% precision to pull that dose out, which means that, intending to pull 10 mL out of that vial, we actually pulled somewhere between 9.6 and 10.4 mL out of that vial, which means that we have delivered somewhere between 830.4 mg and 1.245 grams of Cefazolin out of the vial.
- Our total tolerance stack therefore has a range of ~-17% to +25%, all of which is completely beyond our control.
- As noted previously, that 50 mL IVPB bag actually contains somewhere between 53 and 63 mL. So by the time we add in the between 9.6 and 10.4 mL of drug and shoved it into the bag, we now have between ~63 and 74 mL in the bag. What do we tell the nurse to infuse? What is the concentration in the bag? It is somewhere between ~13.2 and ~17 mg/mL (as opposed to the 20 mg/mL the labeling would lead one to expect).
If we apply gravimetrics to the preparation process, and those gravimetrics correct the concentration computed for the vial based on detecting the addition of 9.4 mL rather than 9.6 mL, it is still only an approximation since we cannot know what was really in the vial in the first place, by a surprisingly wide margin. The range that was 830 mg to 1245 mg is reduced to 870 mg to 1200 mg. Since many gravimetric systems have an acceptable error in the ±5% range, there’s not much of this process they can ameliorate. What they can do is present evidence that suitable mass of something was transferred from one container to another. And that’s valuable.
Applying this same analysis to liquid medications (with a variance of ±10%), we are looking at roughly a ±14% variance (removes reconstitution variance).
Note that human error as not been accounted for at all in this process. Again, most of our measurements are likely somewhere between this range of values, but producing a dose at the edges of the range is quite possible. Note also that we have been living with this amount of variance for the over 40 years I have been in practice. If there is evidence that making the process more accurate produces better outcomes, I have not seen it.
So where does that leave us?
There are things we think we do now that we can’t really do.
There’s a reason that children’s’ hospital pharmacies prepare dilutions; they create doses they can actually measure with acceptable accuracy.
The fact that you can move the plunger in a syringe to a particular mark on its scale does not necessarily mean you are accurately measuring that amount, especially at the lower end of the scale.
Given all the inherent error in the systems we operate, the best we can do is to operate those systems as best we can. This means:
- Setting practical boundaries on what is acceptable.
- Follow the labeling of the products in their preparation
- Prepare dilutions as needed to enable accurate measurements
- Always using the smallest syringe that will measure the entire dose.
- Measuring to, rather than between the lines, rounding values as appropriate. Interpolation cannot add to the accuracy.
- Using tools, like gravimetrics, to tell us whether we managed to get within acceptable limits and to demonstrate appropriate performance during the compounding process.
The other thing worth noting is that if and when we get cost-effective end-product testing in the IV room, we are likely to have to set some pretty broad standards for what is acceptable, because there is a lot of variance we just cannot control.
What do you think?
Dennis A. Tribble, PharmD, FASHP
Ormond Beach, FL
DATdoc@aol.com
The opinions in this blog are my own, and are not necessarily those of my employer or of ASHP